 In 1790, Jean-Antoine Chaptal proposed the name nitrogene from nitre, the French name for potassium nitrate, and Greek -gene meaning to produce. Antoine Lavoisier proposed the name azote from Greek meaning no life, as animals and flames died in pure nitrogen, and whilst this was adopted in other languages, only the chemical term azo- was adopted in English, as did the name pnictogen (Greek for producing suffocation) for the group to which nitrogen belongs. Rutherford did not identify it as an element and several other contemporary scientists also investigated it and proposed the names burnt air and phlogisticated air. Even though 78% of the Earth’s atmosphere is nitrogen gas, it was only discovered in 1772 by Daniel Rutherford who called it noxious air. Display:Ĭarbon fibre, anthracite coal, charcoal, graphite brushes from a motor and graphite electrodes, low density polyethylene and different allotropes of carbon: diamonds (loose), diamond-tipped drill bit, graphite, fullerenes (carbon nanotubes), graphene (in solution) Nitrogen – N – element 7 History:Īmmonia, nitric acid and nitrate salts were known, in some cases, since ancient times. Carbon has a crucial biological role and all plant and animal products contain carbon. Carbon fibres are used in woven materials. 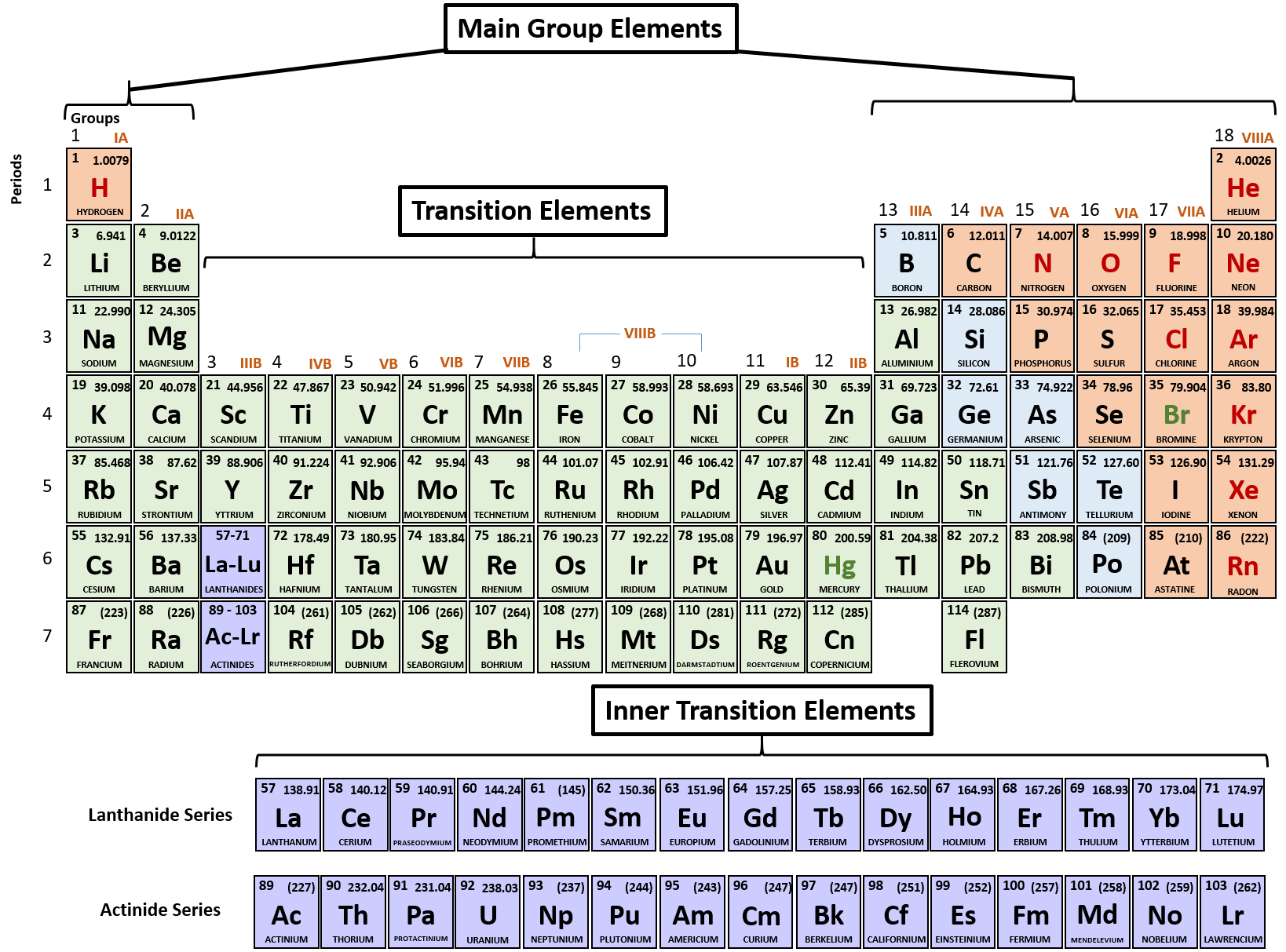
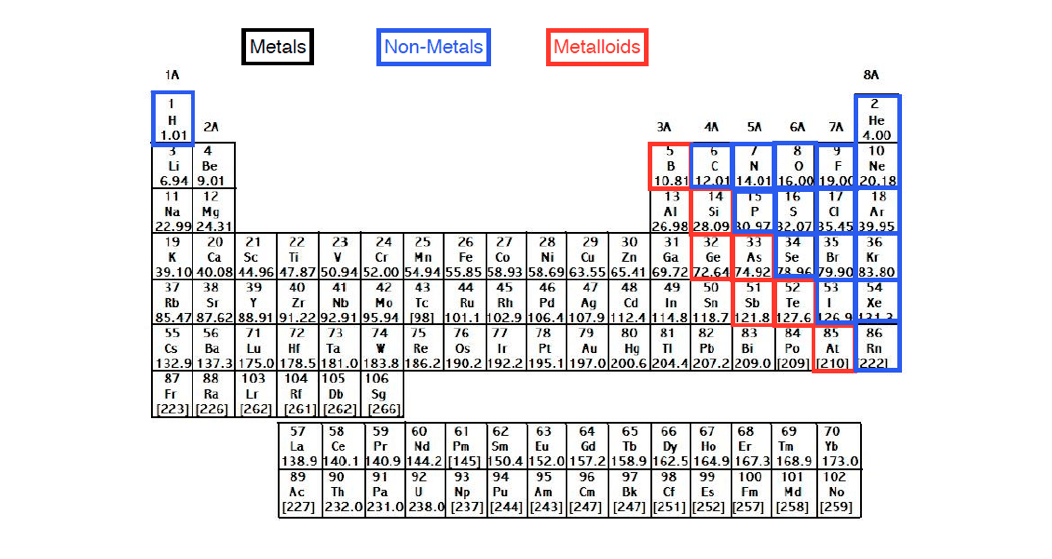
Uses:Ĭarbon’s main industrial uses are as a fuel (coal, oil, and gas) and for making plastics, it is combined with iron to make steel, and diamonds are used in cutting and drilling tools. They proposed the name carbone after the Latin carbo meaning charcoal. Vandermonde did the same with graphite, previously believed to be a type of lead. Similarly, in 1786, Claude Louis Berthollet, Gaspard Monge, and C. In 1772, Antoine Lavoisier showed that diamonds and charcoal produced the same reaction products when burnt. Display:Ĭarbon was known since prehistoric times in the form of charcoal, however it was not known that diamonds and graphite are the same element. Boron compounds such as borax and boric acid are used domestically as cleaning products, insecticides, and food additives amongst other uses. Uses:īoron is used as an additive in glass, particularly fiberglass and borosilicate glass, and in ceramics. The name boron was adopted by analogy with carbon. In 1808 boron was isolated independently by Joseph Louis Gay-Loussac and Louis Jacques Thenard, and Humphry Davy who proposed the name boracium, after the mineral borax. Together with groups 17 and 18, these elements form the p-block.Īlthough boron-containing minerals were known historically, boron compounds were not used widely prior to the 19th century when the mineral borax became widely available. The lighter group 15 elements can form anions with a -3 charge, and the lighter group 16 elements can form anions with a -2 charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
